Bacteriocin producers from traditional food products
Received on 22 August 2006, accepted on 2 May 2007
Résumé
Sélection de bactéries lactiques productrices de bactériocine à partir d'aliments fermentés traditionnels d'origine sénégalaise. Un total de 220 souches de bactéries lactiques isolées de produits fermentés traditionnels d'origine sénégalaise a été testé pour détecter une production de bactériocine. Deux espèces bactériennes, Lactococcus lactis subsp. lactis et Enterococcus faecium, ont été identifiées parmi 12 colonies bactériocinogènes, sur la base d'analyses phénotypiques et du séquençage du gène codant pour l'ARNr 16S. Les bactériocines produites par les deux souches développent une activité bactéricide contre Listeria monocytogenes et Bacillus coagulans tandis que seule la souche Lactococcus lactis est active contre Bacillus cereus. Les souches de Lactococcus lactis subsp. lactis bactériocinogènes ont été détectées dans différents types de produits alimentaires indiquant un potentiel de croissance élevé de cette souche dans divers environnements microbiologiques. Les séquences partielles du gène codant pour l'ARNr 16S des deux souches ont été enregistrées dans la base de données internationale “ Genbank ” sous les numéros AY971748 pour Lactococcus lactis subsp. lactis (CWBI-B1410) et AY971749 pour Enterococcus faecium (appelée CWBI-B1411). La souche Lactococcus lactis subsp. lactis a été sélectionnée pour l'identification et l'application de la bactériocine à la conservation des denrées alimentaires.
Abstract
A total of 220 strains of LAB isolated from 32 samples of traditional fermented food from Senegal were screened for bacteriocin production. Two bacteriocin producers, Lactococcus lactis subsp. lactis and Enterococcus faecium, were identified from 12 bacteriocin-producing isolates on the basis of phenotypic analyses and 16S rDNA sequence. Both bacteriocins produced by new isolates show antimicrobial activity against Listeria monocytogenes and Bacillus coagulans whereas only that produced by Lactococcus lactis has an activity against Bacillus cereus. Bacteriocin-producing Lactococcus lactis strains were found in a variety of traditional foods indicating a high potential of growth of this strain in variable ecological complex environment. Partial 16S rDNA of the two bacteriocin producers obtained in this study has been registered to Genbank databases under the accession number AY971748 for Lactococcus lactis subsp. lactis (named CWBI-B1410) and AY971749 for Enterococcus faecium (named CWBI-B1411). The new bacteriocin-producing Lactococcus lactis subsp. lactis strain has been selected for identification and application of the bacteriocin to food preservation.
1. Introduction
1Lactic acid bacteria (LAB) are the biological basis for the production of a great multitude of fermented foods (Lasagno et al., 2002). The most important contribution of these bacteria to fermented products is to preserve the nutritive qualities of the raw material and inhibit the growth of spoilage and pathogenic bacteria (Matilla-Sandholm et al., 1999). This inhibition may be due to the production of many metabolites such as organic acids (lactic and acetic acid), hydrogen peroxide, diacetyl and bacteriocins (Ennahar et al., 2000 ; Lasagno et al., 2002). Some bacteriocins kill only bacteria belonging to the same species as producer whereas other bacteriocins kill a broad range of Gram positive bacteria (Conventry et al., 1997 ; Ennahar et al., 2000 ; Mc Auliffe et al., 2001 ; Garneau et al., 2002). The incorporation of these compounds as biopreservative ingredient into model food has been shown to be effective in the control of pathogenic and spoilage micro-organisms (O'Sullivan et al., 2002). They have attracted considerable interest in recent years and several works have focused on the isolation and development of new strains of bacteriocin-producing bacteria. The detection rate of bac+ strains from LAB isolates can be as low as 0.2% and therefore needs a large number of isolates from food sources (Conventry et al., 1997).
2The preservation of foods by lactic fermentation has a long history of use in Africa. The multitude of products can be an appropriate ecological habitat for holding wild strains of LAB capable of producing bacteriocin. The aim of the current study was to select bacteriocin-producing LAB from such products in order to use these proteinaceous inhibitors to improve the microbial quality and safety of foods.
2. Materials and methods
2.1. Culture and media
3MRS1 agar (MRS with 0.1% glucose, and 50 µg/ml of cycloheximide) and M17m agar (0.5% glucose and 50 µg/ml cycloheximide) were utilised for isolation of bacteria from food sources. Different food-borne pathogens and Gram+ bacteria including Escherichia coli, Salmonella infantis, Salmonella typhimurium, Staphylococcus aureus, Listeria monocytogenes and Lactobacillus curvatus from CWBI collection were used as sensitive. Strains of Lactobacillus curvatus are found to be the most suitable indicator for the quantification of antimicrobial effects of all bacteriocins investigated in both agar and broth system (Conventry et al., 1997).
2.2. Food sources
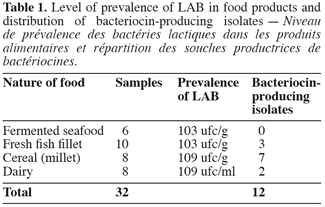
4Strains were isolated from a total of 32 samples of traditional foods from Senegal (Table 1).
2.3. Detection of antimicrobial activity
5Lactic acid bacteria were isolated from samples, by direct plating on MRS1 or M17m. A 10% (w/v) food sample in diluent [0.1% (w/v) peptone] was homogenized and 10-fold serially diluted. Plates of serial dilution in MRS1 and M17m media were incubated anaerobically (BD, BBL Campypak microaeroplilic systems Envelopes, Sparks, USA) for 48 h at 30°C. Plates providing a total of 300 colonies were overlaid with a set of 6 indicators and incubated at 37°C for 12 h. Colonies producing zones of growth inhibition in the indicator lawn were isolated from within the agar, inoculated into broth media (MRS1 or M17m) and incubated for 24 h at 30°C. Culture supernatant was prepared as follows: an overnight culture of each isolate was centrifuged at 8,000 r.p.m. The resulting supernatant was neutralized (pH 6.5) with NaOH 5N, sterilized by filtering with acrodisc (pore size 0.22 µm) and assayed for the presence of an inhibitor in the broth following the Agar well diffusion assay (WDA) technique (Barefoot et al., 1983) as follows. Molten agar was first seeded with indicator organism (110 µl of overnight culture per 20 ml of agar) in sterile Petri dishes, and after solidification, dried for 15 min. under flow hood. Wells of uniform diameter (6 mm) were bored in the agar. Aliquots (60 µl) of the cell-free supernatant (CFS) were dispensed in wells, and plates were incubated overnight at 37°C. Inhibition of growth was determined by an area of inhibition surrounding each agar well.
2.4. Bacteriocin assay
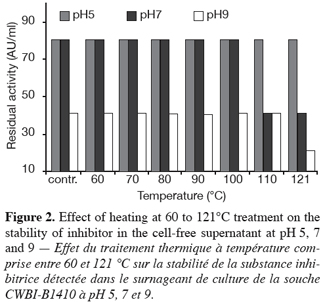
6To confirm the bacteriocin effect, catalase (65 UI/ml) was added to CFS and the technique was repeated. The effect of various enzymes and heat treatment of CFS activity were also investigated. Units and MIC (Minimum inhibitory concentration): the activity present in the neutralized (pH 6.5) cell-free supernatant of producing cultures was determined by twofold serial dilution of the supernatant in sterile phosphate buffer pH 6 (Barefoot et al., 1983). Activity units per milliliter (AU/ml) were determined as the inverse of the last dilution at which growth inhibition was still detectable following the agar WDA. To determine the effects of enzymatic treatments, samples (180 µl of twice the minimum inhibitory concentration corresponding to the supernatant from the cell-free culture) were incubated with 20 µl portion of following enzyme solutions, P3911 (16.6 UI/ml), type XIV (7.9 UI/ml), type XVIII (0.66 UI/ml), proteinase K (59.2 UI/ml), chymotrypsin (700 UI/ml) at 37°C for 1 h 30 min. (Jack et al., 1996) and the residual activity was measured following the WDA. Positive controls were incubated with 20 µl of 50 mM phosphate buffer (pH 6.5). To determine the effect of temperature and pH on the stability of the inhibitor sample corresponding to a dilution of ¼ of neutralized cell-free supernatant (pH 6.5) in 50 mM phosphate buffer at pH 5, 7 and 9, and aliquots of each were subsequently heated to 60, 70, 80, 90, 100, 110 and 121°C for 10 min. (Ryan et al., 1996). The remaining activity was assayed and compared to activity at each pH prior to heat treatment.
2.5. Bacterial identification
7Selected isolates were examined microscopically for cellular morphology and Gram stain phenotype. Catalase activity was tested by spotting colonies with 3% hydrogen peroxide. Fermentation of different sugars was determined by API 50 CHL (Biomerieux). PCR was used to amplify the 16S rRNA gene of bacteriocin-producing strains. The 16S rDNA sequence was determined by direct sequencing. Total DNA was isolated by using Wizard genomic DNA purification kit (Promega, Madison, USA). Primers used for PCR and DNA sequencing are presented in table 2. The PCR amplification was performed with the primer pair SPO/SP6 targeted against regions of 16S rDNA (Ventura et al., 2001). Amplification of DNA was performed in a Mastercycler personal thermal cycler (Eppendorf). PCR conditions included a hot start at 96°C (5 min.), 25 cycles consisting of hybridation at 50°C (1 min), polymerisation at 72°C (2 min.), denaturation at 96°C (1 min) and a final extension at 72°C (10 min.). PCR products were resolved by electrophoresis in 1% (w/v) agarose gel and visualized by ethidium bromide (1 µl/10 ml) staining. 16S rDNA PCR amplicons were purified following the microcon YM-100 kit (Bedford, MA, USA) and sequenced using the Big Dye Terminator V3.0 kit as specified by the supplier with primers described in table 2 while automated sequencing of both strands of the PCR products was done on an ABI 3100 automated gene sequencer (ABI, Forster, USA). The sequences obtained (350–500 bp) were then assembled in silico (Vector NTI) using overlapping zones between the various sequences to form the contiguous sequence. Phylogenetic analysis was realised by an alignment of sequence consensus of the 16S rDNA genes collected in an international database (Genebank). The results were then expressed in percentage of homology between the submitted sequence and the sequences resulting from the database.

3. Results and discussion
3.1. Detection of antimicrobial activity and bacteriocin assay
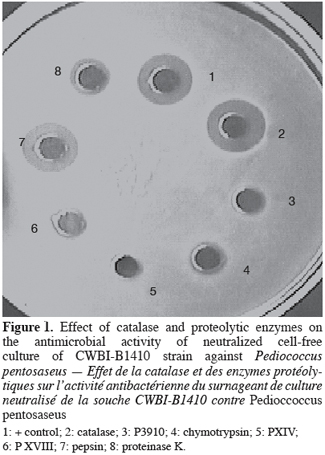
8A total of about 70000 colonies isolated from food samples were examined for detection of antibacterial activity against a set of 6 indicators. A total of 340 (0.6% detection rate) colonies producing a growth inhibition area in the indicator lawn were recorded. Staphylococcus aureus demonstrated the highest detection rate among indicator bacteria. Two hundred and twenty colonies which displayed antibacterial activity against the indicator lawn were randomly isolated and purified; 20 of these strains produced antibacterial activity in the neutralized cell-free supernatant, whereas only 12 confirmed the activity when the CFS was treated with catalase (65 UI/ml). The activity was either completely or partially inactivated by proteolytic enzymes (Figure 1) but was resistant to heat (Figure 2). These results demonstrated that antimicrobial compounds produced by our 12 isolates were heat stable protein or peptide indicating bacteriocin-like substances. Four food samples (12.5% incidence rate) yielded bacteriocin-producing strains. The distribution of bacteriocin producers in food samples is presented in table 2. Our observations could be correlated with similar studies reported these last years. Garver et al. (1993) have reported 13% of products yielding bacteriocin-producers by direct plating and 21% by enrichment while Conventry et al. (1997) obtained 43% by direct plating and 46% by enrichment. Lasagno et al. (2002) identified two bacteriocin-producers among 206 isolates selected on the basis of the inhibition of Lactobacillus plantarum by the CFS using the WDA. Four of the twelve bacteriocin-producing isolates obtained (designated CWBI-B1410, CWBI-B1411, CWBI-B1427 and CWBI-B1428) were selected for further study on the basis of their food source, indicator used for detection, activity against seven indicators and stability of activity upon repeated subcultures.
3.2. Inhibitory spectra
9The sensivity of 33 bacterial strains from different genera to the bacteriocin-like substances produced by the four selected isolates are presented in table 3. Neutralised cell-free supernatant from CWBI-B1410, CWBI-B1427 and CWBI-B1428 isolates demonstrated similar spectra of activity broader than the one produced by the CWBI-1411 isolate. This one showed a reasonably diverse spectrum. All bacteriocins produced by the four isolates showed antimicrobial activity against Bacillus coagulans involved in food spoilage, and the food-poisoning bacterium Listeria monocytogenes, whereas only bacteriocins produced by CWBI-B1410, CWBI-B1427 and CWBI-B1428 isolates inhibited Bacillus cereus.

3.3. Bacteriocin activity
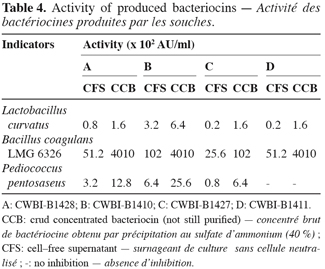
10Activity units per ml (AU/ml) of bacteriocins was determined following WDA assay and presented in table 4. Bacillus coagulans showed the more sensivity to bacteriocins. The level of activity against this strain, (104 to 105 AU/ml), can be correlated with similar studies reported these last years. Flynn et al. (2002) have reported 106 AU/ml, using a bacteriocin produced by Lactobacillus salivarius subsp. salivarius, whereas Garcia et al. (2003) reported 105 AU/ml with enterocin EJ97 produced by Enterococcus feacalis EJ97. Bacteriocins produced by CWBI-B1410, CWBI-B1427 and CWBI-B1428 isolates also demonstrated an activity of 103 AU/ml against Pediococcus pentosaseus whereas that produced by CWBI-B1411 is more active against Listeria monocytogenes (data not shown). CWBI-B1410 showed the highest production of bacteriocin.
3.4. Identification of bacteriocin-producers
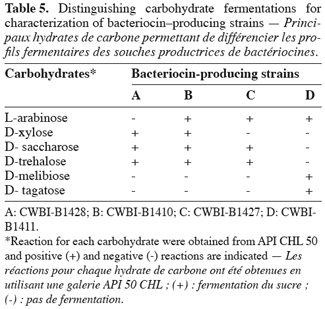
11Bacteriocin-producing isolates were catalase negative, and Gram positif cocci (CWBI-B1410, CWBI-B1427 and CWBI-B1428) or ovoid (CWBI-B1411). Fermentation of different carbohydrates was performed using the API 50 CHL system (API Biomerieux). 22 reactions (sugar fermentation) were determined and 6 of them provided a mean of discriminating them (Table 5). Identification made by the API database correlation indicated that all the four strains were Lactococcus lactis subsp. lactis. However, a low percentage of similarity (91%) was obtained for CWBI-B1411 (Table 6).

12Nucleotide sequences of 16S rDNA of the four bacteriocin-producing isolates were carried out to confirm or infirm biochemical species identification. The determined 16S rDNA sequences of isolates were compared directly with the Genebank database. A high level of similarity of 16S ribosomal DNA nucleotide sequences (99% of matches) of CWBI-B1410, CWBI-B1427 and CWBI-B1428 strains was observed with the sequences of Lactococcus lactis subps. lactis strains whereas the sequence of CWBI-B1411 strain matched best with that of Enterococcus faecium strains (Table 6). The closest matches for CWBI-B1411 strain and Enterococcus faecium were different from the identification determined by API method (Table 6). However, we prioritized genetic identification because of it accordance with morphology analysis, and the low percentage of similarity obtained by biochemical analysis which could be due to the inadequacy of API CHL 50 for well identification of Enterococcus strains. Partial 16S rDNA of the two bacteriocin-producing isolates Lactococcus lactis subsp. lactis (CWBI-B1410) and Enterococcus faecium (CWBI-B1411) have been registered to Genebank databases respectively under the accession numbers AY971748 and AY971749. Due to the high amount of bacteriocin produced in the culture supernatant, the Lactococcus lactis subsp. lactis strain (named CWBI-B1410) was selected for further investigations.
4. Conclusion
13Traditional fermented foods from Senegal provide an appropriate ecological habitat for wild bacteriocin-producing LAB. Bacteriocin producers mainly belong to Lactococcus lactis subsp. lactis group. Bacteriocin-producing Lactococcus lactis strains were found in a variety of fermented products indicating a high potential of growth of this strain in different ecological complex environment. The produced bacteriocins showed a broad spectrum of activity including spoilage microorganisms and pathogens associated with food such as Bacillus coagulans, Listeria monocytogenes and Bacillus cereus. These results show the potential usefulness of these bacteriocins justifying a more in depth investigation for their identification and application as food biopreservatives.
14Acknowledgments
15We are grateful to Belgian University Development Centre (CUD) who financially supported this research which was realized in both, the Centre Wallon de Biologie Industrielle (CWBI)/ Gembloux Agricultural University,Belgium and the Ecole Supérieure Polytechnique (ESP)/ University of Dakar/Senegal.
Bibliographie
Barefoot SF., Klaenhammer TR. (1983). Detection anbactivity of lacticin B, a bacteriocin produced by Lactobacillus acidophilus. Appl. Environ. Microbiol. 45, p. 1808–1815.
Conventry MJ., Gordon JB., Wilcock A., Harmark K., Davidson BE., Hickey MW., Hillier AJ., Wan J. (1997). Detection of bacteriocin of LAB isolated from food and comparison with pediocin and nisin. J. Appl. Microbiol. 83, p. 248–258.
Ennahar S., Sashihara T., Sonomoto K., Ishizaki A. (2000). Class IIa bacteriocins: biosynthesis, structure and activity. FEMS Microbiol. Rev. 24, p. 85–106.
Flynn S., Sideren DV., Thornton GM., Holo H., Nes IF., Collins JK. (2002). Characterisation of the genetic locus responsible for the production of ABP-118, a novel bacteriocin produced by the probiotic bacterium Lactobacillus salivarius subsp. salivarius UCC 118. Microbiology 148, p. 973–984.
Garcia MT., Ben Omar N., Lucas R., Pérez-Pulido R., Castro A., Grande MJ., Martínez-Cañamero M., Gálvez A. (2003). Antimicrobial activity of Enterocin EJ.97 on B. coagulans CECT12. Food Microbiol. 20, p. 533–536.
Garneau S., Martin NI., Vederas JC. (2002). Two-peptide bacteriocins produced by lactic acid bacteria. Biochimie 84, p. 577–592.
Garver KI., Muriana PM. (1993). Detection, identification and characterization of bacteriocin producing lactic acid bacteria from retail food products. Int. J. Food Microbiol. 19, p. 241–258.
Jack RW., Wan J., Gordon J., Harmark K., Davidson BE., Hillier AJ., Wettenhall REH., Hickey MW., Coventry MJ. (1996). Characterisation of the chemical and antimicrobial properties of piscicolin 126, a bacteriocin produced by Carnobacterium piscicola JG 126. Appl. Environ. Microbiol. 62, p. 2897–2903.
Lasagno M., Beoletto V., Sesma F., Raya R., Font De Valdez G., Eraso A. (2002). Selection of bacteriocin producer strains of lactic acid bacteria from a dairy environment. Microbiologia 25, p. 37–44.
Matilla-Sandholm T., Mättö J., Saarela M. (1999). LAB with health claim-interactions and interference with gastrointestinal flora. Int. Dairy J. 9, p. 25–35.
McAuliffe O., Ross RP., Hill C. (2001). Lantibiotics: structure, biosynthesis and mode of action. FEMS Microbiol. Rev. 25, p. 285–308.
O’Sullivan L., Ross RP., Hill C. (2002). Potential of bacteriocin-producing lactic acid bacteria for improvements in food safety and quality. Biochimie 84, p. 593–604.
Ryan MP., Rea MC., Hill C., Ross RP. (1996). An application in Cheddar Cheese Manufacture for a strain of Lactococcus lactis producing a Novel broad-spectrum bacteriocin, lacticin 3147. Appl. Environ. Microbiol. 62, p. 612–619.
Ventura M., Elli M., Reniero R., Zink R. (2001). Molecular microbial analysis of bifidobacterium isolates from different environments by the species-specific amplified ribosomal DNA restriction analysis (ARDRA). FEMS Microbiol. Ecol. 36, p. 113–121.

