- Accueil
- volume 12 (2008)
- numéro 2
- Bovine colostrum as a natural growth promoter for newly weaned piglets: a review
Visualisation(s): 11405 (37 ULiège)
Téléchargement(s): 417 (6 ULiège)
Bovine colostrum as a natural growth promoter for newly weaned piglets: a review

Notes de la rédaction
Received on July 23, 2007, accepted on December 20, 2007
Résumé
Utilisation du colostrum bovin en tant que promoteur de croissance naturel chez le porcelet au sevrage : synthèse bibliographique. L'objectif de cette synthèse bibliographique est de présenter le potentiel du colostrum bovin en tant que promoteur de croissance chez le porcelet au sevrage. Les conséquences du sevrage sur les performances de croissance, le tube digestif, le métabolisme et le système endocrinien des porcelets sont décrites dans la première partie de cette synthèse. Une seconde partie est dédiée au colostrum bovin, avec une description des promoteurs de croissance et des facteurs antimicrobiens contenus dans celui-ci. Finalement, les effets d'éléments spécifiques du colostrum ou de fractions de colostrum mesurés sur des porcelets au sevrage sont présentés. Ils indiquent clairement le potentiel du colostrum bovin pour réduire les pertes de performances associées au sevrage du porcelet.
Abstract
The aim of this review is to present the potential of bovine colostrum as growth promoter in piglet-weaner diet. The consequences of weaning on the growth performance, on the gastro-intestinal tract and on the metabolic and endocrine systems of the piglet are described in the first part of this review. The second part is dedicated to bovine colostrum, with a description of the actions due to its main growth promoters and antimicrobial factors. Finally, the reported effects of colostrum specific components or colostrum fractions on the growth performance and on the structure and function of the gastro-intestinal tract of piglets in the early postweaning period are presented. They show clearly the potential of bovine colostrum to reduce the growth-check related to the weaning of the piglet.
Table des matières
1. Introduction
1Weaning can be regarded as one of the most critical periods in the modern-day pork production cycle. In addition to mother-young separation, weaning involves abrupt and profound modifications of the environment, feeding habits and social interactions when litters of piglets are mixed. These changes contribute to the post-weaning (PW) " growth check ", intimately associated with a range of intestinal and immunological alterations (Pluske et al., 1997).
2Over the last decades, antibiotic growth promoters have been used in weaner diets to reduce the production penalty associated to weaning. However, increased bacterial resistance to antibiotics led the European Union to implement a full ban on in-feed antibiotics from January 2006. Efficient alternatives, therefore, have to be found to conform to this policy change. One possible alternative could be bovine colostrum powder.
3Bovine colostrum is a commercially available co-product of the dairy industry. More than a source of nutrients, colostrum also contains several biologically active molecules that are essential for specific functions (Pakkanen et al., 1997). The most important bioactive components in colostrum include growth and antimicrobial factors. Growth factors promote the growth and development of the newborn, while antimicrobial factors provide passive immunity and protect against infections during the first weeks of life. Growth factors include insulin-like growth factors-I and -II (IGF-I and IGF-II), transforming growth factor-β1 and -β2 (TGF-β1 and TGF-β2) and epidermal growth factor (EGF). Antimicrobial factors include lactoferrin, lysozyme, lactoperoxidase, immunoglobulins (Igs) and cytokines (IL-1β, IL-6, TNF-α, IFN-γ and IL-1ra).
4The aim of this review is to present the potential of bovine colostrum as growth promoter in piglet-weaner diet. The review is divided into three sections. A first section describes the consequences of weaning on the growth performance, on the gastro-intestinal tract and on the metabolic and endocrine systems of the piglet. A second section is dedicated to the bovine colostrum, with a description of the actions due to its main growth promoters and antimicrobial factors. A third section presents the reported effects of bovine specific components or bovine fractions on the growth performance and on the structure and function of the gastro-intestinal tract of piglets in the early PW period.
2. Piglet weaning
5In natural or semi-natural conditions weaning is a progressive process taking place around 12 to 17 weeks of age in pig (Stolba et al., 1989; Boe, 1991). In modern pig husbandry, weaning occurs abruptly at the age of 3 to 4 weeks (Mormède et al., 2003), inducing numerous stressors to piglets. All these factors contribute to a range of intestinal and immunological alterations.
2.1. Consequences of weaning on growth
6Behavioural studies reported by Brooks (1999) indicate that, although the majority of pigs start to eat solid feed within 5 h after weaning, some of them take up to 54 h before eating their first meal. Thereafter, metabolisable energy (ME) intake gradually increases at a rate of 100-120 kJ ME.kg-0,75 per day. Regardless of the age of weaning, the level of ME intake attained at the end of the 1st PW week ranges between 700 and 800 kJ.kg-0,75, which accounts for 60-70% of the pre-weaning milk ME intake. This reduction of ME intake is due to the transition from milk to a less digestible solid feed, resulting in a critical period of underfeeding (Le Dividich et al., 2000). This abrupt reduction in voluntary feed intake immediately PW results in the so-called " weaning growth check " and its severity has a major impact on subsequent performance (Tokach et al., 1992; Azain, 1993). On the first day PW, piglets loose from 100 to 250 g of body weight (BW) (Le Dividich et al., 2000). This loss of BW is dependent on the age of weaning. Carroll et al. (1998) measured a reduction of average daily BW gain (ADG) of 0.09 kg.d-1 for piglets weaned at 2 weeks of age while the reduction of the ADG for piglets weaned at 3 weeks was of 0.06 kg.d-1. Growth rate subsequently returned to pre-weaning levels within 9 and 6 days for the piglets weaned at 2 and 3 weeks of age, respectively.
2.2. Gastro-intestinal modifications induced by weaning
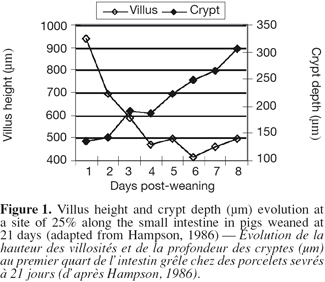
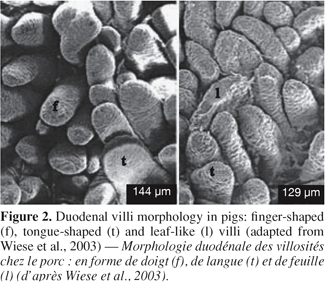
7Morphological changes. The small intestine and its mucosa lose 20-30% of their relative weight during the first 2 days PW (Lallès et al., 2004). This reduction is associated with a villous atrophy followed by a crypt hyperplasia described by a lot of authors (Figure 1) (see Pluske et al., 1997 for a review). These changes are more conspicuous when weaning occurs earlier at 14 days rather than later at 28 days of age. Cera et al. (1988) reported additionally to those changes, a reduction in the length of microvilli 3 to 7 days after weaning. Villous atrophy at weaning is caused by an increased rate of cell loss which induces an increase in crypt-cell production and, hence, an increase in crypt depth. As a result of these changes in villous height and crypt depth after weaning, the villous height/crypt depth ratio in weaned pig is markedly reduced compared to unweaned animals. These structural changes are variable along the small intestine. Hampson (1986) reported that the change in villous height was greater at the proximal small intestine in piglets weaned on day 21, but the change in crypt depth was greatest at the distal small intestine. Gu et al. (2002) confirmed a greater change in duodenum morphology compared to the other parts of the small intestine. Associated with the reduction in villous height and the increase in crypt depth, the morphology of the villi also changes from long finger-shaped before weaning to leaf- or tongue-like structures after weaning (Cera et al., 1988; Wiese et al., 2003) (Figure 2).
8Digestive and absorptive capacity modifications.
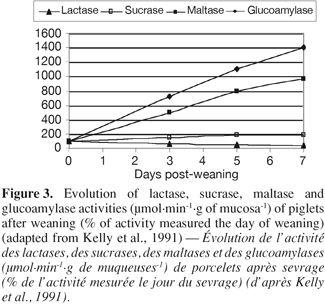
9– Enzymatic activity: Concomitant to the structural changes, there are marked alterations in intestinal functions following weaning. According to Gu et al. (2002), the villous atrophy and the crypt hyperplasia reduce the number of mature enterocytes and lead to a reduction in the activity of the brush-border enzymes, which is related to the limited digestive and absorptive area or specific digestive and absorptive capacity of the small intestine, respectively. Hampson et al. (1986) and Pié et al. (2004) reported rapid reductions in the specific activities of lactase and sucrase during the first 4-5 days after weaning, with a greater loss in lactase than in sucrase activity, probably due to the more apical distribution of lactase activity along the villous (Tsuboi et al., 1981 and 1985, cited by Pluske et al., 1997). Miller et al. (1986) reported that the specific activities of sucrase, lactase and isomaltase fell by at least 50% of during the first 5 days after weaning in pigs weaned at 28 or 42 days of age. On the other hand, the activities of maltase and glucoamylase doubled from the day 3 to the day 7 PW (Figure 3) (Kelly et al., 1991). Increases in these polysaccharidases are likely the result of substrate induction.
10– Absorptive activity: In several studies, the decrease in villous height, the increase in crypt depth, and the loss of digestive enzyme activity after weaning, coincided with a reduced ability in intestinal absorption of sugar (D-xylose: Hampson et al., 1986), amino acids (alanine: Miller et al., 1986) and glucose and electrolytes (Nabuurs et al., 1994). Nevertheless, some other studies failed to detect a significant reduction in the ability of sugar absorption (Kelly et al., 1991).
11Microflora modifications. Whilst the piglet is suckling, the dominant bacteria within the stomach and small intestine tend to be Lactobacilli and Streptococci, which are well adapted to utilise substrate from the milk diet. Following weaning, the period of starvation and then the consumption of the new solid diet result in altered availability of specific microbial substrate all along the digestive tract. The intestinal structural and functional changes may also be responsible for modifications to the mass, composition and complexity of the intestinal microflora, leaving the pig more susceptible to overgrowth with potentially disease-causing pathogenic bacteria (Hopwood et al., 2003). But these modifications may also be responsible for structural and functional changes. For example, Mroz et al. (2003) showed that enteric infections after weaning further depress intestinal enzyme activities.
12Jensen (1998) quantified changes in bacterial populations that occur in the small and large intestine of piglets weaned at 28 d of age. In the small intestine, the previously predominant Lactobacilli decreased in number during the first week after weaning, whilst the total number of bacteria and the proportion of coliforms, Escherichia coli in particular, increased. Following this period of perturbation of the intestinal microflora, it subsequently re-stabilises. Franklin et al. (2002) observed also an effect of the age of weaning of the piglets on the microflora modifications, younger piglets having a higher variation of their microflora than older (17 d vs. 24 d of age).
13Immunological modifications. The intestinal epithelium provides an extensive and complex interface between the piglet's immune system and its environment, which must function simultaneously to absorb digested nutrients and provide a barrier against a vast array of ingested antigens. In addition to its barrier function, the epithelium also functions in surveillance. It can signal the onset of the host innate and acquired immune response through the production of cytokines and chemokines that are crucial for the recruitment and activation of neutrophils, macrophages, T and B cells, and dendritic cells (King et al., 2003; Pié et al., 2004).
14– Immune cells: McCracken et al. (1999) observed that piglets weaned at 21 days of age had an increase in jejunal lamina propria CD4+ and CD8+ T lymphocytes within 2 and 7 days, respectively, after weaning. They reported also an increased expression of the active form of the matrix metalloproteinase stromelysin in jejunal explants during the initial 7 days after weaning and a decrease in jejunal expression of MHC class I and II mRNA.
15Vega-Lopez et al. (1995) measured, 4 days after weaning at 21 days of age, an increase in the CD2+ and macrophage cells population in proximal small intestinal villi and only in the CD2+ cells population in crypts, but no changes were observed in cells populating the distal small intestine. Similar results were reported by Solano-Aguilar et al. (2001) who observed gradual changes in CD4+ and CD8+ T cells, monocytes, granulocytes and macrophages in the month after weaning.
16– Cytokines: The first observations on cytokine responses to weaning were reported by McCracken et al. (1995). They observed a transient increase of plasma IL-1 during the first 2 days PW.
17More recently, with the development of the RT-PCR analysis, it was demonstrated that weaning in piglets is associated with an early regulation of inflammatory cytokines in the gut. Pié et al. (2004) reported an intestinal up-regulation of the pro-inflammatory cytokines IL-1β, IL-6 and TNF-α during the first 2 days after weaning. While between day 2 and day 8 PW, gene expression returned to pre-weaning levels, except for TNF-α in the colon. This early gene up-regulation of pro-inflammatory cytokines probably contributes to early functional disorders favouring diarrhoea (Lallès et al., 2004).
2.3. Metabolic changes around weaning
18Due to the weaning stressors and the low feed intake, newly weaned piglets are often in negative energy balance until 3 to 5 days PW (Le Dividich et al., 2000). However, the nitrogen balance remains positive (Bruininx et al., 2002). It follows that the energy required for maintenance, physical activity and protein deposition implies necessary a loss of body fat. The body fat catabolism conduces to a transient increase in plasma free fatty acids concentrations, whereas lipogenesis is marginal (Fenton et al., 1985). This catabolism continues until an ADG of 200g.d-1 is reached and it takes 3 to 6 weeks until the initial body fat content is recovered, depending on body weight and age at weaning. This decrease in body fat induces a reduced body thermal insulation which, associated to low feed intake, results in a transient increase in the lower critical temperature of the piglets from 22-23°C at weaning to 26-28°C during the first week PW (Le Dividich et al., 1994).
2.4. Effects of weaning on the hormonal status
19The onset of weaning results in some quite profound hormonal changes, although it is difficult to separate causes and effects. Many of the changes are in response to the social and nutritional stress associated to weaning, but there are also overlying developmental changes that likely occur independently of the weaning process (Dunshea, 2003).
20Somatotropic hormones. Weaning results in an increase in the level of blood GH and a decrease of blood IGF-I concentrations (White et al., 1991; Carroll et al., 1998; Matteri et al., 2000). These observations may be related to the transient underfeeding period at weaning as fasting for 36 h resulted in the same response of the somatotropic axis (Kasser et al., 1981). Moreover, Carroll et al. (1998) showed that circulating IGF-I did not recover to pre-weaning value until 7 to 10 days PW, a period roughly corresponding with the time required by the piglets to attain their pre-weaning ME intake.
21Hypothalamic-pituitary axis hormones. Regardless of the age of weaning, a transient increase in plasma cortical concentration and in urinary cortisol excretion is observed during the 2 first days PW (Carroll et al., 1998; Colson et al., 2006). This increased cortisol secretion could reflect both the weaning stress and stimulation of gluconeogenesis associated with the low PW feed intake (Le Dividich et al., 2000).
22The actual role of the catecholamine system in the regulation of the weaned pig metabolism is still unclear. Colson et al. (2006) and Hay et al. (2001) studied the effect of weaning on catecholamine production and observed decreased levels of urinary adrenaline and noradrenaline. Hay et al. (2001) interpreted this as a consequence of food intake deficit. However, according to Dunshea (2003), underfeeding would lead to an increase in circulating catecholamine to favour the mobilisation of energy stores during the early PW period.
23Thyroid hormones. Plasma concentration of thyroid hormones (3.5.3'-triiodothyronine, T3 and thyroxine, T4) are reported to respond to feeding level, decreasing with the reduction in feed intake or with malnourishment (Le Dividich et al., 2000). Carroll et al. (1998) observed unaltered levels of T3 and T4 after weaning, but measured a decline of those two hormones after a change in the diets.
3. Bovine colostrum
3.1. Definition and composition
24Colostrum is the lacteal secretion directly after parturition common to all mammals and essential for development and immune status for newborn (Scammell, 2001). More than a source of nutrients such as proteins, carbohydrates, fat, vitamins and minerals, colostrum also contains several biologically active molecules that are essential for specific functions (Pakkanen et al., 1997). The most important bioactive components in colostrum include growth factors and antimicrobial factors. Some of these factors are also present in regular milk, but in much lower concentrations (e.g., typically 1:100 to 1:1000 of what is found in colostrum) (Maher, 2000). Table 1 compares the concentrations of the main components of bovine colostrum and regular milk.

3.2. Growth promoters
25IGF-I and -II and their binding proteins (BP). The most abundant growth factors of bovine colostrum are IGF-I and -II. Both IGF-I and -II are single chain polypeptides with 70 and 67 amino acid residues and molecular weights of about 7.6 and 7.5 kDa, respectively. The primary structures of IGF-I and -II are highly conserved across species and have identical sequences in pigs, humans and cattle (Xu et al., 2000). They stimulate cell growth and differentiation and are proposed to act both as endocrine hormones through the blood and, locally, as paracrine and autocrine growth factors. IGF-I is biologically more potent than IGF-II (Jones et al., 1995).
26Six IGFBP have been identified and cloned. The detectable level and rank of specific IGFBP in bovine mammary secretions are IGFBP-3 > IGFBP-2 ≈ IGFBP-4 > IGFBP-5 (Blum et al., 2002). Those binding proteins are involved in the regulation and the coordination of biological activities of the IGF-I and -II (Hwa et al., 1999).
27In biological fluids, IGF-I is usually bound to its binding proteins (IGFBP), which have also been detected in bovine milk. IGF-I appears in mature milk mainly in the bound form (85-90%), but in the first milkings postpartum the free form of IGF-I predominates (73%). The slightly acidic pH (6.3) of the colostral secretion is correlated with an increased proportion of the free IGF-I (Einspanier et al., 1991).
28Epidermal Growth Factor (EGF) receptor ligand family. The polypeptides of this family have the common property of binding to the EGF receptor (a 175 kDa cell surface glycoprotein with tyrosine kinase activity). The most important members of this family are EGF itself and TGF-α (Barnard et al., 1995).
29EGF is a 6 kDa peptide, composed of 53 amino acids. The peptide is highly homologous among species and elicits similar effects across species (Odle et al., 1996). Colostral EGF may play a role:
30– in the prevention of bacterial translocation,
31– in the stimulation of gut growth in suckling neonates by playing an important role in cell differentiation rather than cell proliferation and in stimulating mucus secretion (Schweiger et al., 2003).
32TGF-α is a 6 kDa peptide, composed of 50 amino acids and shares about 30% sequence identity with EGF. It may play a complementary role to that of TGF-β (see below) in controlling the balance between cell proliferation and differentiation in the intestinal epithelium (Playford et al., 2000).
33Transforming growth factor βeta (TGF-β). Three isoforms of TGF-β (TGF-β1, β2 and β3) are known. Those forma are homodimeric proteins with a molecular weight of approximately 25 kDa (Jin et al., 1991). TGF-β1 and β2 have been isolated from bovine colostrum, with a predominance of the β2 form (85-95%) (Elfstrand et al., 2002).
34TGF-β is a highly pleiotropic growth factor with several different types of function. It stimulates proliferation of some cells, especially in connective tissue, whereas it acts as a growth inhibitor of some other cells, such as lymphocytes and epithelial cells. TGF-β plays an important role in embryogenesis, tissue repair, formation of bone cartilage, and in the control of the immune system (Tripathi et al., 2006). During injury or disease, it acts in concert with EGF to stimulate cell proliferation (Border et al., 1995).
3.3. Antimicrobial factors
35The colostral antimicrobial factors contribute to the protection of the neonate against infectious diseases, which is crucial for its survival. These factors may be classified in three groups according to their action:
36– specific antimicrobial factors (immunoglobulins),
37– non-specific antimicrobial factors (lactoferrin, lactoperoxidase and lysozyme),
38– factors which have both specific and non-specific activities (cytokines).
39Lactoferrin. Lactoferrin is an 80 kDa iron-binding glycoprotein present in colostrum, milk, and to a lesser extent in other exocrine fluids such as tears. It is a member of the transferring family of non-heme iron-binding protein, characterised by their unique anion requirement for binding of iron (Viljoen, 1995).
40Lactoferrin has been shown to inhibit the growth of several microbes, including Escherichia coli, Salmonella typhimurium, Shigella dysenteria, Listeria monocytogenes, Streptococcus mutans, Bacillus subtilis (Pakkanen et al., 1997). In fact, lactoferrin exhibits both bacteriostatic and bactericidal activity against a range of microorganisms. The bacteriostatic activity is related to the high iron binding affinity of the protein that deprives iron-requiring bacteria of this essential growth nutrient. The bactericidal mechanism is related to its ability to cause release of lipopolysaccharide (LPS) molecules from the outer membrane of the Gram-negative bacteria. Recent results suggest that a cationic domain at the N-terminus of lactoferrin is responsible for its bactericidal properties. This domain is distinct from the amino acids involved in iron binding, indicating that the bactericidal activity of lactoferrin is distinct from metal chelation (Conneely, 2001).
41In addition to its antimicrobial activity, it has been proposed that lactoferrin plays an important role in iron uptake in the intestine and in the activation of phagocytes and immune responses. Receptors for lactoferrin are found on intestinal tissues, monocytes, macrophages, neutrophils, lymphocytes, platelets and on some bacteria (Viljoen, 1995; Hoek et al., 1997). By its function of binding free iron, lactoferrin may act as an antioxidant, protecting the immune cells against free radicals produced by themselves in areas of inflammation or infection (Britigan et al., 1994). Moreover, by its ability to bind and neutralize LPS, lactoferrin will reduce production of cytokines in response to inflammation or infection (Cohen et al., 1992).
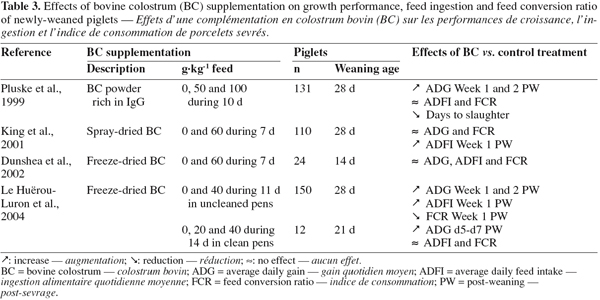
42Lactoperoxidase. Lactoperoxidase is a basic glycoprotein of 78 kDa containing a heme-group with Fe3+. It catalyses the oxidation of thiocyanate (SCN-) in the presence of hydrogen peroxide (H2O2), producing a toxic intermediary oxidation product. This product inhibit bacterial metabolism via the oxidation of essential sulphydryl groups in microbial enzymes and other proteins (Pruitt et al., 1985).
43According to Kussendrager et al. (2000), lactoperoxidase has a bactericidal activity against Gram-negative and a bacteriostatic activity against Gram-positive bacteria. This difference in sensitivity can probably be explained by the difference in cell wall structure and their different properties. Additionally, some viruses, including polioviruses, appear to be sensitive to the toxic effects of lactoperoxidase. Next to that antimicrobial and antiviral activity, degradation of various carcinogens and protection of animal cells against peroxidative effects have been reported (Kussendrager et al., 2000).
44Lysozyme. Lysozyme, originally described by Alexander Fleming in 1922, is a lytic enzyme of 14.3 kDa. The natural substrate of this enzyme is the peptidoglycan layer of the bacterial cell wall and its degradation results in lysis of the bacteria (Reiter, 1978). Due to the difference of the outer membrane structure between Gram-negative and Gram-positive bacteria, the lysozyme action is more intensive on Gram-positive bacteria, leading to the death of the bacteria, while it does not adversely affect the viability of Gram-negative bacteria (Ibrahim et al., 1994). Over the last 15 years, several authors have proposed a novel antibacterial mechanism of action of lysozyme, independent of its enzymatic activity. The precise mechanism remains unknown, but it seems evident that bactericidal activity depends on the passage through the outer membrane (Masschalck et al., 2001).
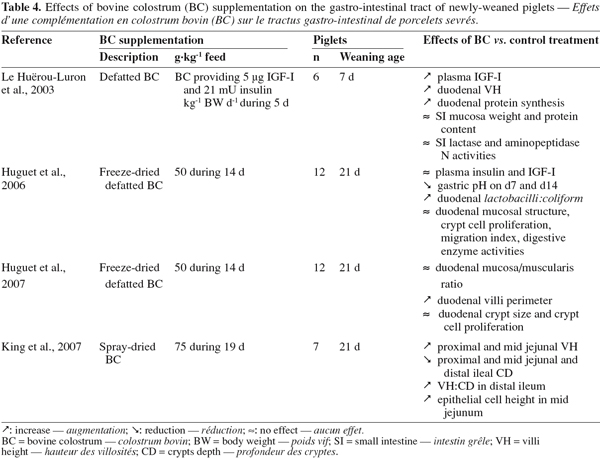
45A particularity of the lysozyme is its interaction with other factors present in the colostrum. It activates partly lactoperoxidase by forming a complex with it (Hulea et al., 1989). In presence of lactoferrin, the antimicrobial activity of lysozyme against E. coli is also enhanced as lactoferrin damages the outer membrane of Gram-negative bacteria and the organism becomes susceptible to lysozyme (Yamauchi et al., 1993). Finally, it works also in synergy with IgA and complement factors against E. coli (Hill et al., 1974).
46Immunoglobulins (Igs). Igs are glycoproteins constituted by four amino acid chains: two identical light chains (23 kD) and two identical heavy chains (50-70 kD). According to the structure of their heavy chains, they can be divided into five classes: IgG, IgA, IgM, IgE and IgD.
47Igs are present in very high concentrations in colostrum. They represent 70-80% of the total protein contents in colostrum (up to 100 g.l-1 in bovine colostrum) (Elfstrand et al., 2002), whereas in mature milk Igs account for only 1-2% of the protein (Table 1).
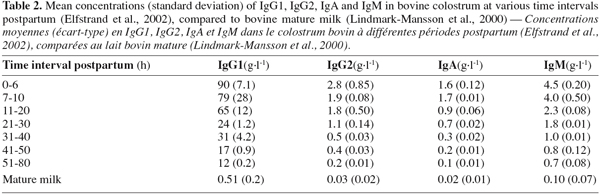
48Three classes of Igs are present in bovine colostrum: IgG, IgA and IgM. The major Igs present in bovine colostrum are IgG, among which 95% belong to the subclass IgG1 and 5% to the IgG2. Table 2 shows the evolution of the Igs concentrations during the 80 first hours postpartum in colostrum and in mature milk. It shows that the concentrations of the individual Igs decline at different rates over the time it takes to reach the concentration in mature milk (Elfstrand et al., 2002).
49In the neonates the colostral Igs are transferred from the lumen of the intestine into the circulation through a non-selective macromolecular transport system across the small intestinal epithelium. This non-selective absorption occurs only within about 24-36 h after birth and provides the transmission of passive immunity from the cow to its calf (Pakkanen et al., 1997). However, it has been shown that older animals can absorb Igs, but larger quantities of these antibodies are required for an effective transport (Maher, 2000).
50All Igs exhibit one or more effector function in addition to antigen binding. Whereas one part of an antibody binds to antigen, other parts interact with other elements. The immunological function mediated by the Igs depends on the Ig class. The most important action of IgG antibodies is the activation of complement-mediated bacteriolytic reactions. Another vital function is their ability to increase the recognition and phagocytosis of bacteria by leucocytes. IgM antibodies are considerably more efficient than IgG in regards of the above activities, especially complement-mediated lysis. IgA, in contrast, does not fix complement or opposing bacteria, but agglutinates antigens, neutralises viruses and bacterial toxins, and prevents the adhesion of enteropathogenic bacteria to mucosal epithelial cells. Moreover, this Ig is present in bovine colostrum under a secretory form (sIgA) that makes it resistant to the activities of proteolytic digestive enzymes (Korhonen et al., 2000).
51Cytokines. Colostrum and milk contain many cytokines, including IL-1β, IL-6, TNF-α, IFN-γ and IL-1ra (Hagiwara et al., 2000). The concentrations of these cytokines are really low, but they are active from picomolar to nanomolar concentrations.
52Cytokines are small peptide molecules that are important mediators in the regulation of the immune and inflammatory responses. In general, cytokines do not regulate normal cellular homeostasis, but alter cellular metabolism during times of perturbation, e.g. in response to inflammation. In the newborn, these factors play an important role in combination with the ingested maternal Ig and the non-specific antibacterial components in colostrum (Playford et al., 2000). Of particular interest is the role of cytokines as major regulators of epithelial cell growth and development, including intestinal inflammation and epithelial restitution following mucosal damage (Elson et al., 1994).
4. Use of bovine colostrum in pig production at weaning
53Bovine colostrum and its main components have already been largely studied on neonates and in vitro. In this review, we will focus on the results obtained on weaned or adult pigs when they are existent. Their particularity, compared to neonates, is the gut closure, suggesting that absorption of molecules of bovine colostrum is no more possible. Nevertheless, according to Jensen et al. (2001), the presence of bioreactive components in colostrum may be responsible for an enhanced uptake of molecules after closure by inducing changes in brush border enzyme activities. Moreover, in the case of Igs, Stirling et al. (2005) demonstrated that the absorption of bovine or porcine IgG colostrum is possible in adult animals (more than 4-weeks-old animals).
4.1. Effects of specific components
54Growth promoters. According to Pluske et al. (1997), an opportunity may exist to enhance growth and development through supplementation of the newly weaned pig with exogenous growth factors. As argued by Dunshea et al. (1995), this is particularly pertinent in the case of the weaned piglet since (a) its gut is relatively "immature" at weaning, (b) the pig suffers a growth check, and (c) the gut of the newly weaned pig is often colonised by enteropathogenic bacteria.
55– IGF-I: Oral IGF-I and IGF-II at pharmacologic doses can stimulate cellular proliferation in the gastro-intestinal tract in newborn pigs (Burrin et al., 1996; Xu et al., 1996a). Intestinal tissues in porcine neonates apparently do not have the capacity to absorb significant amounts of ingested IGF-I and transport this peptide intact to the circulation (Xu et al., 1996b; Donovan et al., 1997). Insulin-like growth factors are anabolic and possible differentiation-inducing factors for intestinal epithelium of newborns, suggesting possible applications of recombinant IGF and IGF analogues for repair of damaged gastro-intestinal tissues (Simmen et al., 1998). However, Marion et al. (2002) showed that a weaning diet supplemented with IGF-I induced an increase in plasma concentrations in IGF-I, but had no effect on the intestinal structure of 7 days old weaned piglets. These controversial observations need further investigations.
56– EGF: Exogenous EGF administered either orally or systemically stimulates gastro-intestinal tissue growth in weaned animals. Read et al. (1986) reported that inclusion of EGF (200 µg.kg-1) in the diet significantly accelerated intestinal growth in weaning rats following 50% removal of the small intestine. However, no studies on the effects of EGF on the growth of the intestinal tissues of weaned piglets were found.
57In addition to its growth-promoting effect, EGF appears to be able to modulate the enterocyte differentiation during the transition phase from maternal milk to solid food in weaned piglets (Schweiger et al., 2003). In 21-day old newly weaned piglets, orally administered EGF (372 µg.d-1) increased jejunal lactase (by 77%) and sucrase (by 97%) specific activities measured after 3 days of feeding (Jaeger et al., 1990). Moreover, Kingsnorth et al. (1990) also reported increased tensile strength of gastric wounds in 20-kg pigs after 5 days of intra-peritoneal infusion of EGF (0.5 µg.kg-1.d-1). These results suggest that supplementation with EGF may aid in the recovery of traumatised gastric and intestinal tissues. Playford et al. (2000) proposed that EGF acts as a " luminal surveillance peptide " in the adult gut, readily available to stimulate the repair process at sites of injury.
58Antimicrobial factors
59– Immunoglobulins: Effects of bovine milk Igs on weaned piglets' performance have been demonstrated. Leibbrant et al. (1987) showed that milk-derived Igs added to milk replacers increase growth performance following gut closure in early-weaned pigs. Pierce et al. (2005) attributed this improvement to the continuous source of Igs until the pig is capable of synthesising its own Igs. Moreover, Stirling et al. (2005) demonstrated the presence of porcine Fc receptors that transferred orally delivered (bovine or porcine) IgG into the blood supply and concluded that this receptor has the potential to deliver protein antigens to the pig immune system.
60In the gastro-intestinal tract, immuno-supplementation with bovine immunoglobulin in the form of specific antibody has been shown to be effective against various enteric diseases. Although bovine colostrum contains Igs to neutralise enteric pathogens, the titer of Igs present is considered by some researchers too low to afford protection against specific infectious organisms. This limitation is overcome by the production of hyperimmune bovine colostrum. In trials on infants, it has been successfully shown that specific antibodies in bovine milk are effective against both enteropathogenic and enterotoxigenic Escherichia coli, cryptosporidium, Helicobacter pylori, Rotavirus and Shigella flexneri (See Tripathi et al., 2006 for a review).
61No studies with hyperimmune colostrum in weaning piglets have been found. However Schaller et al. (1992) demonstrated in a gnotobiotic piglet model that both viral shedding and diarrhoea were effectively reduced or eliminated in a dose-dependent manner, as a result of feeding Igs preparations containing antibodies specific for human rotavirus strains. Moreover, Mroz et al. (1999) showed that passive protection of piglets against PW collibacillosis could be provided by egg IgG immunised against enterotoxigenic strains of Escherichia coli.
62– Lactoferrin: Wang et al. (2007) studied the effects of lactoferrin (1 g.kg-1 basal diet) on growth performance, intestinal microflora and morphology of weaning pigs. They observed an increase in growth performance (increase in ADG by 34% and in ADFI by 17%), a better-feed efficiency (decrease in F/G by 12.8%). Effects on the intestinal microflora were a reduction of the total viable counts of Escherichia coli and Salmonella, and an enrichment of the Lactobacillus and Bifidobacterium. The villus height was increased and crypt depth decreased at the small intestinal mucosa, as compared with the basal diet. They concluded that the use of lactoferrin as an additive would be a good method of defending weaned pigs from infections and weanling stress.
4.2. Effects of colostrum supplementation
63The administration of bovine colostrum presents two main advantages due to its specific components: – it allows the synergic effects of different components such as described above for the lysozyme for example,
64– it has been reported that porcine milk contains potent inhibitory activity against trypsin and chymotrypsin, and prevents growth factors, such as EGF and IGF-I, hydrolysis in neonatal and newly-weaned pig intestinal fluids (Shen et al., 1996; 2000) suggesting that colostrum, the natural carrier of milk-borne growth factors, may protect the peptides from gastro-intestinal luminal digestion.
65Studies of the effects of bovine colostrum fractions on growth performance, feed ingestion and feed conversion ratio of newly weaned piglets are presented in table 3.
66Except for Dunshea et al. (2002), the three other studies presented in the table 3 showed an increase in ADG during the first week PW. Pluske et al. (1999) observed an increase of 40% and 80% of the ADG with a starter diet supplemented with 50 and 100 g of bovine colostrum extract per kg of feed, respectively. King et al. (2001) observed an increase of 20% (P > 0.05) with 60 g of bovine colostrum per kilo of feed and Le Huërou-Luron et al. (2004) showed an increase of the ADG up to 110% with a starter diet supplemented with 40 g of bovine colostrum per kilo of feed. In all these studies, the authors explained the increase in ADG by an increase in the ADFI (+ 10 and + 25% for Pluske et al., 1999, + 25% for King et al., 2001 and Le Huërou-Luron et al., 2004).
67Interestingly, Le Huërou-Luron et al. (2004) observed in an " unclean " environment a reduction of 10% of the FCR, which was not observed in the other studies performed in cleaned rooms. They concluded that bovine colostrum had more effects in bad environmental conditions (uncleaned pens) than in a clean environment, as it was observed with plasmatic proteins (van Dijk et al., 2001). This suggests that the positive response of bovine colostrum might result from an improvement in the sanitary status of the colostrum-treated piglets by a direct effect of the colostrum on gut health.
68Effects of bovine colostrum observed on the gastro-intestinal tract of newly weaned piglets are presented in table 4. Bovine colostrum helped to preserve intestinal integrity after weaning, especially in the duodenum where bovine colostrum maintained higher villi (+ 10 to 20%) (Le Huërou-Luron et al., 2003; Huguet et al., 2007; King et al., 2007) and reduced crypt depth (- 6%) (King et al., 2007) the first week PW compared to a control treatment. Moreover, Le Huërou-Luron et al. (2003) showed also an increase in the duodenal protein synthesis. All these observations show the beneficial effect of bovine colostrum to preserve the intestinal mucosa integrity.
69An effect of bovine colostrum-supplemented diets was also observed on the intestinal microflora by Huguet et al. (2006), who showed an improved lactobacilli:coliform ratio in bovine colostrum treated weaned piglets, compared to weaned piglets receiving a control diet without colostrum (1.63 vs. 1.19). This effect was mainly caused by lower coliforms counts whereas lactobacilli counts were identical between colostrum treated and control piglets. This observation confirms the higher effects of bovine colostrum reported by Le Huërou-Luron et al. (2004) in an " unclean " environment and indicates the positive contribution of bovine colostrum in the improvement of gut health around weaning in piglets.
70Another effect of importance observed by Huguet et al. (2006) in the gastro-intestinal tract is the lower gastric pH in bovine colostrum treated piglets. This strengthened the effects of colostral antibacterial components and may also be at the origin of the Coliform population reduction, the later being less resistant to low pH than Lactobacilli.
71Finally, Le Huërou-Luron et al. (2003) reported an increase in plasma IGF-I (+ 33%) in weaned piglets receiving bovine colostrum. They explained this observation by an increase of the synthesis and secretion of IGF-I under the action of growth promoters contained in bovine colostrum, but they could not determine which one.
5. Conclusion and perspectives
72According to the results presented on the use of bovine colostrum in piglets diet PW, it appears clearly that bovine colostrum supplementation improves the growth performance and the sanitary status of piglets during the early PW period. These beneficial effects are explained by both an increase in feed intake level the first days and a likely direct effect on gut health (preservation of gut integrity and reduction of the coliform population).
73Those results are really encouraging but many works are still necessary to understand the mechanism of action of the bovine colostrum and to make this natural product technically and economically competitive compared to other alternatives (probiotics, prebiotics, enzymes, organic acids, plant extracts, etc.) in the weaner diet.
74Firstly, the action of bovine colostrum on the structure of the gut has been studied but no information is available on its local action on the gut associated lymphoid tissue (GALT) of newly weaning piglets. This is of first importance to evaluate its effect on the local immune system of the piglet.
75Secondly, the effects on the systemic immune system should also be studied to evaluate the effect of bovine colostrum to extend our knowledge about the action of bovine colostrum to the other organs of the weaned pig.
76Finally, future investigations on lower doses of bovine colostrum supplementation in the weaner diet are necessary to reduce the costs of its use. Indeed, except the study of Le Huërou-Luron et al. (2004) where 20 and 40 g of bovine colostrum per kilo of feed were used, all the other cited authors incorporated from 5 to 10% of colostrum in their starter diets, thereby increasing too excessively the price of the diet in our modern pork production systems.
77List of abbreviations
78ADFI: average daily feed intake
79ADG: average daily gain
80BW: body weight
81EGF: epidermal growth factor
82FCR: feed conversion ratio
83GALT: gut associated lymphoid tissue
84Ig: immunoglobulin
85IGF: insulin-like growth factor
86IGFBP: IGF binding protein
87IL: interleukine
88LPS: lipopolysaccharide
89ME: metabolisable energy
90PW: post-weaning
91T3: 5.3'-triiodothyronine
92T4: thyroxine
93TGF: transforming growth factor
Bibliographie
Azain M.J., 1993. Impact of starter diet on nursery performance. Swine Report n°86. Athens, USA: University of Georgia, 49-54.
Barnard J.A. et al., 1995. Epidermal growth factor-related peptides and their relevance to gastro-intestinal pathophysiology. Gastroenterology, 108, 564-580.
Blum J.W. & Hammon H., 2000. Colostrum effects on the gastro-intestinal tract, and on nutritional, endocrine and metabolic parameters in neonatal calves. Livest. Prod. Sci., 66, 151-159.
Blum J.W. & Baumrucker C.R., 2002. Colostral and milk insulin-like growth factors and related substances: mammary gland and neonatal (intestinal and systemic) targets. Domest. Anim. Endocr., 23, 101-110.
Boe K., 1991. The process of weaning in pigs: when the sow decides. Appl. Anim. Behav. Sci., 30, 47-59.
Border W.A. & Noble N.A., 1995. Targeting TGF-β for treatment of disease. Nat. Med., 1, 1000-1001.
Britigan B.E., Serody J.S. & Cohen M.S., 1994. The role of lactoferrin as an anti-inflammatory molecule. In: Hutchens T.W., Rumball S.V. & Lonnerdal B. Lactoferrin: structure and function, advances in experimental medicine and biology. New York, USA: Plenum Press, 143-156.
Brooks P.H., 1999. Strategies and methods for the allocation of food and water in the post-weaning period. In: 50th Annual meeting of the European Association for Animal Production, August 22-26, 1999. Zurich, Switzerland: EAAP.
Bruininx E.M.A.M. et al., 2002. A prolonged photoperiod improves feed intake and energy metabolism of weanling pigs. J. Anim. Sci., 80, 1736-1745.
Burrin D.G. et al., 1996. Orally administered IGF-I increases intestinal mucosal growth in formula-fed neonatal pigs. Am. J. Physiol., 270, R1085-R1091.
Carroll J.A., Veum T.L. & Matteri R.L., 1998. Endocrine responses to weaning and changes in post-weaning diet in the young pig. Domest. Anim. Endocr., 15, 183-194.
Cera K.R. et al., 1988. Effect of age, weaning and postweaning diet on small intestinal growth and jejunal morphology in young swine. J. Anim. Sci., 66, 574-584.
Cohen M.S. et al., 1992. Interaction of lactoferrin and lipopolysaccharide (LPS): effects on the antioxidant property of lactoferrin and the ability of LPS to prime human neutrophils for enhanced superoxide formation. J. Infect. Dis., 166, 1375-1378.
Colson V., Orgeur P., Foury A. & Mormède P., 2006. Consequences of weaning piglets at 21 and 28 days on growth, behaviour and hormonal responses. Appl. Anim. Behav. Sci., 98, 70-88.
Conneely O.M., 2001. Antiinflammatory activities of lactoferrin: review. J. Am. Coll. Nutr., 20, 389S-395S.
Donovan S.M., Chao J.C., Zijlstra R.T. & Odle J., 1997. Orally administered iodinated recombinant human insulin-like growth factor-I (125I-rhIGF-I) is poorly absorbed by the newborn piglet. J. Pediatr. Gastroenterol. Nutr., 24, 174-182.
Dunshea F.R., 2003. Metabolic and endocrine changes around weaning. In: Pluske J.R., Le Dividich J. & Verstegen M.W.A. Weaning the pig. Concepts and consequences. Wageningen, the Netherlands: Wageningen Academic Publishers, 61-80.
Dunshea F.R. & Walton P.E., 1995. Potential of exogenous metabolic modifiers for the pig industry. In: Hennessy D.P. & Cranwell P.D. Manipulating pig production V. Werribee, Victoria, Australia: Australasian Pig Science Association, 42-51.
Dunshea F.R. et al., 2002. Diets containing high-quality animal proteins increase growth of early-weaned pigs. Aust. J. Agric. Res., 53, 779-784.
Einspanier R. & Schams D., 1991. Changes in concentrations of insulin-like growth factors 1 and 2 and their binding proteins in human milk: effect of heat treatment on IGF and IGF binding protein stability. J. Dairy Res., 58, 171-178.
Elfstrand L. et al., 2002. Immunoglobulins, growth factors and growth hormone in bovine colostrum and the effects of processing. Int. Dairy J., 12, 879-887.
Elson C.O. & Beagley K.W., 1994. Cytokines and immune mediators. In: Johnson L.R. Physiology of the gastro-intestinal tract. 3rd ed. New York, USA: Raven, 243-266.
Fenton J.P., Roehrig K.L., Mahan D.C. & Corley J.R., 1985. Effect of swine weaning age on body fat and lipogenic activity in liver and adipose tissue. J. Anim. Sci., 60, 190-199.
Franklin M.A., Mathew A.G., Vickers J.R. & Clift R.A., 2002. Characterization of microbial populations and volatile fatty acid concentrations in the jejunum, ileum, and cecum of pigs weaned at 17 vs 24 days of age. J. Anim. Sci., 80, 2904-2910.
Gopal P.K. & Gill H.S., 2000. Oligosaccharides and glycoconjugates in bovine milk and colostrum. Br. J. Nutr., 84(S1), 69-74.
Gu X., Li D. & She R., 2002. Effect of weaning on small intestinal structure and function in the piglet. Arch. Anim. Nutr., 56, 275-286.
Hagiwara K. et al., 2000. Detection of cytokines in bovine colostrum. Vet. Immunol. Immunopathol., 76, 183-190.
Hampson D.J., 1986. Alteration in piglet small intestine structure at weaning. Res. Vet. Sci., 40, 32-40.
Hampson D.J. & Kidder D.E., 1986. Influence of creep feeding and weaning on brush border enzyme activities in the piglet small intestine. Res. Vet. Sci., 40, 24-31.
Hay M.P. et al., 2001. Neuroendocrine consequences of very early weaning in swine. Physiol. Behav., 72, 263-269.
Hill I.R. & Porter P., 1974. Studies of bactericidal activity to Escherichia coli of porcine serum and colostral immunoglobulins and the role of lysozyme with secretory IgA. Immunology, 26, 1239-1250.
Hoek K. et al., 1997. Antibacterial activity of bovine lactoferrin-derived peptides. Antimicrob. Agents Chem., 41, 54-59.
Hopwood D.E. & Hampson D.J., 2003. Interactions between the intestinal microflora, diet and diarrhoea, and their influences on piglet health in the immediate post-weaning period. In: Pluske J.R., Le Dividich J. & Verstegen M.W.A. Weaning the pig. Concepts and consequences. Wageningen, the Netherlands: Wageningen Academic Publishers, 199-218.
Huguet A., Sève B., Le Dividich J. & Le Huërou-Luron I., 2006. Effects of a bovine colostrum-supplemented diet on some gut parameters in weaned piglets. Reprod. Nutr. Dev., 46, 167-178.
Huguet A. et al., 2007. Influence of bovine colostrum on restoration of intestinal mucosa in weaned piglets. Livest. Sci., 108, 20-22.
Hulea S.A., Mogos S. & Matei L., 1989. Interaction of lactoperoxidase with enzymes and immunoglobulins in bovine milk. Biochem. Int., 19, 1173-1181.
Hwa V., Oh Y. & Rosenfeld G., 1999. The insulin-like growth factor-binding protein (IGFBP) superfamily. Endocr. Rev., 20, 761-787.
Ibrahim H.R. et al., 1994. Enhanced bactericidal action of lysozyme to Escherichia coli by inserting a hydrophobic pentapeptide into its C terminus. J. Biol. Chem., 269, 5059-5063.
Jaeger L.A., Lamar C.H., Cline T.R. & Cardona C.J., 1990. Effect of orally administered epidermal growth factor on the jejunal mucosa of weaned piglets. Am. J. Vet. Res., 51, 471-474.
Jensen B.B., 1998. The impact of feed additives on the microbial ecology of the gut in young pigs. J. Anim. Feed Sci., 7, 45-64.
Jensen A.R., Elnif J., Burrin D.G. & Sangild P.T., 2001. Development of intestinal immunoglobulin absorption and enzyme activities in neonatal pigs is diet dependent. J. Nutr., 131, 3259-3265.
Jin Y. et al., 1991. Separation, purification, and sequence identification of TGF-β1 and TGF-β2 from bovine milk. J. Prot. Chem., 10, 3411-3416.
Jones J.I. & Clemmons E.R., 1995. Insulin-like growth factors and their binding proteins: biological actions. Endocr. Rev., 16, 3-34.
Kasser T.R., Martin R.J., Gahagan J.H. & Wangness P.J., 1981. Fasting plasma hormones and metabolites in feral and domestic newborn pigs. J. Anim. Sci., 53, 420-426.
Kelly B.Y.D., Smyth J.A. & McCracken K.J., 1991. Digestive development of the early-weaned pig. 1. Effect of continuous nutrient supply on the development of the digestive tract and on changes in digestive enzyme activity during the first week post-weaning. Br. J. Nutr., 65, 169-180.
King M.R. et al., 2001. Inclusion of colostrum powder and bovine plasma in starter diets increases voluntary feed intake. In: Manipulating Pig Production VIII. Werribee, Victoria, Australia: Australasian Pig Science Association, 213.
King M.R., Kelly D., Morel P.C.H. & Pluske J.R., 2003. Aspects of intestinal immunity in the pig around weaning. In: Pluske J.R., Le Dividich J. & Verstegen M.W.A. Weaning the pig. Concepts and consequences. Wageningen, the Netherlands: Wageningen Academic Publishers, 219-257.
King M.R., Wester T.J. & Morel P.C.H., 2007. The effect of dietary spray-dried bovine colostrum and plasma on the response of pigs to enterotoxigenic E. coli challenge after weaning. Livest. Sci., 108, 292-294.
Kingsnorth A.N., Vowles R. & Nash J.R.G., 1990. Epidermal growth factor increases tensile strength in intestinal wounds in pigs. Br. J. Surg., 77, 409-412.
Korhonen H., 1977. Antimicrobial factors in bovine colostrum. J. Sci. Agric. Soc. Finl., 49, 434-447.
Korhonen H., Marnila P. & Gill H.S., 2000. Milk immunoglobulins and complement factors. Br. J. Nutr., 84(S1), S75-S80.
Kussendrager K.D. & van Hooijdonk A.C.M., 2000. Lactoperoxidase: physico-chemical properties, occurrence, mechanism of action and applications. Br. J. Nutr., 84(S1), S19-S25.
Lallès J.P. et al., 2004. Gut function and dysfunction in young pigs: physiology. Anim. Res., 53, 301-316.
Le Dividich J. & Herpin P., 1994. Effects of climatic conditions on the performance, metabolism and health status of weaned piglets. Livest. Prod. Sci., 38, 79-90.
Le Dividich J. & Sève B., 2000. Effects of underfeeding during the weaning period on growth, metabolism, and hormonal adjustments in the piglet. Domest. Anim. Endocr., 19, 63-74.
Le Huërou-Luron I. et al., 2003. Supplementation of a weaning diet with a bovine colostrum extract minimise post-weaning duodenal structural, but not functional, alterations in 7-d old piglet. In: 9th International symposium on digestive physiology in pigs, vol. 2, May 14-17, 2003, Banff, Alberta, Canada. Edmonton, Canada: University of Alberta, 238-240.
Le Huërou-Luron I. et al., 2004. La supplémentation de l'aliment de sevrage en colostrum bovin améliore l'ingestion et les performances zootechniques chez les porcelets au sevrage. In : 36èmes Journées de la Recherche Porcine, 3-5 février 2004, Paris. Paris : Ed. ITP - INRA, 33-38.
Leibbrant V.D., Kemp R.A. & Crenshaw T.D., 1987. Effect of liquid milk replacer supplementation immediately postweaning on nursery pig performance. J. Anim. Sci., 65(S1), 129 (Abstr.).
Lindmark-Mansson H. et al., 2000. Influence of milk components, somatic cells and supplemental zinc on milk processability. Int. Dairy J., 10, 423-433.
Maher T.J., 2000. Bovine colostrum, continuing education module. Boulder, CO, USA: New Hope Institute of Retailing.
Marion J. et al., 2002. La supplémentation de l'aliment de sevrage en un extrait de colostrum bovin augmente la hauteur des villosités dans le duodénum chez le porcelet sevré. In : 36èmes Journées de la Recherche Porcine, 3-5 février 2004, Paris. Paris : Ed. ITP - INRA, 103-108.
Masschalck B., Van Houdt R., Van Haver E.G.R. & Michiels C.W., 2001. Inactivation of Gram-negative bacteria by lysozyme, denaturated lysozyme, and lysozyme-derived peptides under high hydrostatic pressure. Appl. Environ. Microb., 67, 339-344.
Matteri R.L. et al., 2000. Effects of weaning on somatotropic gene expression and circulating levels of insulin-like growth factor-1 (IGF-1) and IGF-2 in pigs. Domest. Anim. Endocrin., 19, 247-259.
McCracken B.A. et al., 1995. Diet-dependent and diet-independent metabolic responses underlie growth stasis of pigs at weaning. J. Nutr., 125, 2838-2845.
McCracken B.A. et al., 1999. Weaning anorexia may contribute to local inflammation in the piglet small intestine. J. Nutr., 129, 613-619.
Miller B.G., James P.S., Smith M.W. & Bourne F.J., 1986. Effect of weaning on the capacity of pig intestinal villi to digest and absorb nutrients. J. Agric. Sci., 107, 579-589.
Mormède P. & Hay M., 2003. Behavourial changes and adaptations associated with weaning. In: Pluske J.R., Le Dividich J. & Verstegen M.W.A. Weaning the pig: concepts and consequences. Wageningen, the Netherlands: Wageningen Academic Publishers, 53-60.
Mroz Z., Dekker R.A., Koopmans S.J. & Le Huërou-Luron I., 2003. Performance, functional features on the digestive tract and haematological indices in weaned piglets fed antibiotic-free diets and exposed to a vitro-bacterial infection. In: Ball R.O. Proceedings of the 9th International symposium on digestive physiology in pigs, vol. 2, May 14-17, 2003, Banff, Alberta, Canada. Edmonton, Canada: University of Alberta, 180-182.
Mroz Z. et al., 1999. Passive protection of piglets against diarrhoea with specialized egg immunoglobulins (Protimax). In: Cranwell P.D. Manipulating Pig Production VII. Werribee, Victoria, Australia: Australasian Pig Science Association, 238.
Nabuurs M.J.A., Hoogendoorn A. & Van Zijderveld F.G., 1994. Effects of weaning and enterotoxigenic Escherichia coli on net absorption in the small intestine of pigs. Res. Vet. Sci., 56, 379-385.
Odle J., Zijlstra R.T. & Donovam S.M., 1996. Intestinal effects of milkborne growth factors in neonates of agricultural importance. J. Anim. Sci., 74, 2509-2522.
Pakkanen R. & Aalto J., 1997. Growth factors and antimicrobial factors of bovine colostrum. Int. Dairy J., 7, 285-297.
Pié S. et al., 2004. Weaning is associated with an upregulation of expression of inflammatory cytokines in the intestine of piglets. J. Nutr., 134, 641-647.
Pierce J.L. et al., 2005. Effects of spray-dried animal plasma and immunoglobulins on performance of early weaned pigs. J. Anim. Sci., 83, 2876-2885.
Playford R.J., Macdonald C.E. & Johnson W.S., 2000. Colostrum and milk-derived peptide growth factors for the treatment of gastro-intestinal disorders. Am. J. Clin. Nutr., 72, 5-14.
Pluske J.R., Hampson D.J. & Williams I.H., 1997. Factors influencing the structure and function of the small intestine in the weaned pig: a review. Livest. Prod. Sci., 51, 215-236.
Pluske J.R. et al., 1999. A bovine colostrum product in a weaner diet increases growth and reduces days to slaughter. In: Manipulating Pig Production VII. Werribee, Victoria, Australia: Australasian Pig Science Association, 256.
Pruitt K.M. & Reiter B., 1985. Biochemistry of peroxidise system. In: Pruitt K.M. & Tenovuo J. The lactoperoxidase system: chemistry and biology significance. New York, USA: Marcel Dekker, 143-178.
Read L.C. et al., 1986. Is orally-derived epidermal growth beneficial following premature birth or intestinal resection? Endocrinol. Exp., 20, 199-207.
Reiter B., 1978. Reviews of the progress of dairy science: antimicrobial systems in milk. J. Dairy Res., 45, 131-147.
Scammell A.W., 2001. Production and uses of colostrum. Aust. J. Dairy Technol., 56(2), 74-82.
Schaller J.P. et al., 1992. Prevention of human rotavirus-induced diarrhea in gnotobiotic piglets using bovine antibody. J. Infect. Dis., 165(4), 623-630.
Schweiger M., Steffl M. & Amselgruber W.M., 2003. Differential expression of EGF receptor in the pig duodenum during the transition phase from maternal milk to solid food. J. Gastroenterol., 38, 636-642.
Shen W.H. & Xu R.J., 1996. Stability of epidermal growth factor in the gastro-intestinal lumen of suckling and weaned pigs. Life Sci., 59, 197-208.
Shen W.H. & Xu R.J., 2000. Stability of insulin-like growth factor I in the gastro-intestinal lumen in neonatal pigs. J. Pediatr. Gastroenterol. Nutr., 30, 299-304.
Simmen F.A. et al., 1998. The porcine insulin-like growth factor system: at the interface of nutrition, growth and reproduction. J. Nutr., 128, 315S-320S.
Solano-Aguilar G.I. et al., 2001. Characterization of lymphocyte subsets from mucosal tissues in neonatal swine. Dev. Comp. Immunol., 25, 245-263.
Stirling C.M.A. et al., 2005. Characterization of the porcine neonatal FC receptor-potential use for trans-epithelial protein delivery. Immunology, 114, 542-553.
Stolba A. & Wood-Gush D.G.M., 1989. The behaviour of pigs in a semi-natural environment. Anim. Prod., 48, 419-425.
Tokach M.D., Goodbank R.D., Nelssen J.L. & Kats L.J., 1992. Influence of weaning weight and growth during the first week post-weaning on subsequent pig performance. Report of Progress 667, Kansas University Swine Day. Manhattan, Kansas, USA: Kansas University, 15-17.
Tripathi V. &Vashishtha B., 2006. Bioactive compounds of bovine colostrum. Food Rev. Int., 22, 225-244.
Van Dijk A.J. et al., 2001. Growth performance of weanling pigs fed spray-dried animal plasma: a review. Livest. Prod. Sci., 68, 263-274.
Vega-Lopez M.A., Bailey M., Telemo E. & Stokes C.R., 1995. Effect of early weaning on the development of immune cells in the pig small intestine. Vet. Immunol. Immunopathol., 44, 319-327.
Viljoen M., 1995. Lactoferrin: a general review. Haematologica, 80, 252-267.
Wang Y.Z. et al., 2007. Effects of the lactoferrin (LF) on the growth performance, intestinal microflora and morphology of weanling pigs. Anim. Feed Sci. Technol., 135, 263-272.
White M.E. et al., 1991. Effect of weaning at different ages on serum insulin-like growth factor I (IGF-I), IGF binding proteins and serum in vitro mitogenic activity in swine. J. Anim. Sci., 69, 134-145.
Wiese F., Simon O. & Weyrauch K.D., 2003. Morphology of the small intestine of weaned piglets and a novel method for morphometric evaluation. Anat. Histol. Embryol., 32, 102-109.
Xu R.J. & Wang T., 1996. Gastro-intestinal absorption of insulin-like growth factor-I in neonatal pigs. J. Pediatr. Gastroenterol. Nutr., 23, 430-437.
Xu R.J. et al., 1996. Morphological changes in the oesophagus of newborn pigs: effects of age, diet and oral insulin-like growth factor-I (IGF-I) or IGF-II. Reprod. Fertil. Dev., 8, 903-909.
Xu R.J., Wang F. & Zhang S.H., 2000. Postnatal adaptation of the gastro-intestinal tract in neonatal pigs: a possible role of milk-borne growth factors. Livest. Prod. Sci., 66, 95-107.
Yamauchi K., Toimita M., Giehl T.J. & Ellison R.T., 1993. Antibacterial activity of lactoferrin and a pepsin-derived lactoferrin peptide fragment. Infect. Immun., 61, 719-728.
Pour citer cet article
A propos de : Christelle Boudry
Gembloux Agricultural University – FUSAGx. Animal Science Unit. Passage des Déportés, 2. B-5030 Gembloux (Belgium). E-mail: boudry.c@fsagx.ac.be
A propos de : Jean-Paul Dehoux
Catholic University of Louvain. Faculty of Medicine. Experimental Surgery Unit. Avenue Hippocrate, 55/70. B-1200 Brussels (Belgium).
A propos de : Daniel Portetelle
Gembloux Agricultural University – FUSAGx. Animal and Microbial Biology Unit. Avenue Maréchal Juin, 6. B-5030 Gembloux (Belgium).
A propos de : André Buldgen
Gembloux Agricultural University – FUSAGx. Animal Science Unit. Passage des Déportés, 2. B-5030 Gembloux (Belgium).






